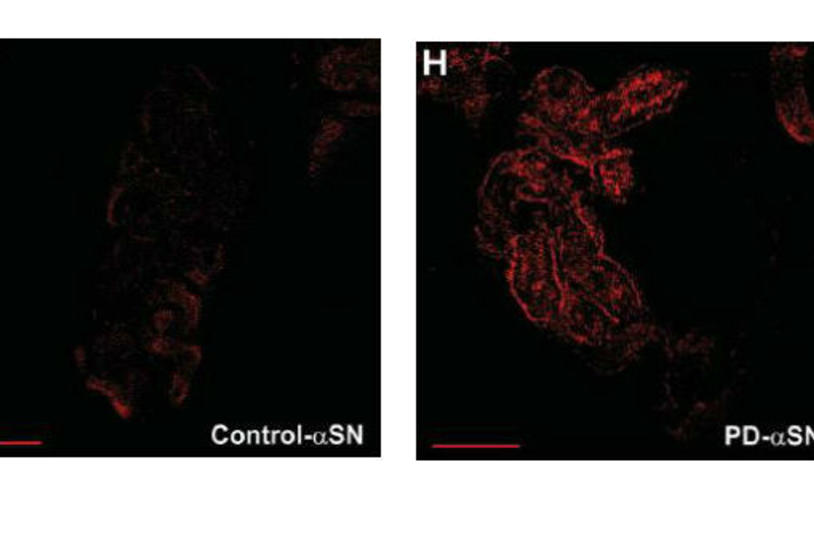
Research has found more alpha-synuclein in the skin of PD patients (right) compared to controls. Image courtesy of Neurology.
Alpha-synuclein shoulders a lot of blame in Parkinson’s disease (PD). This protein clumps in the cells of all Parkinson’s patients, leading to cell dysfunction and cell death. But not all alpha-synuclein may be bad. Scientists do not yet know the normal function of alpha-synuclein, but it is likely that only certain species form the toxic aggregates seen in Parkinson’s.
Proteins occur in different shapes and structures thanks to splicing and modulations. When they interact with other compounds, proteins are changed slightly, resulting in different species of the same base protein.
A new funding program from The Michael J. Fox Foundation will support multi-institute teams to look for different species of alpha-synuclein in clinical samples and determine which play a role in the toxic PD clumps.
“Alpha-synuclein is our most promising target for a drug that could stop or prevent Parkinson’s,” said Todd Sherer, PhD, MJFF CEO. “Understanding what species of this protein are involved in the disease would accelerate development of a disease-modifying therapy.”
Researchers are already measuring alpha-synuclein levels in different parts of the body.
The Parkinson’s Progression Markers Initiative, the landmark biomarker study sponsored by MJFF, is looking at alpha-synuclein in cerebrospinal fluid. MJFF-funded researchers are investigating levels of this protein in the salivary gland and the skin, among other regions.
Understanding which species of alpha-synuclein is toxic could lead researchers to develop more precise drugs that could prevent or break up the protein clumps. A more specified biomarker test of disease risk, onset or progression could allow for faster testing of those disease-modifying therapies, too.
The new funding program will support teams to look at alpha-synuclein in the central nervous system (brain tissue and spinal fluid) and in the peripheral nervous system (blood, plasma, other tissue).
Read more about the Foundation’s research into alpha-synuclein.
