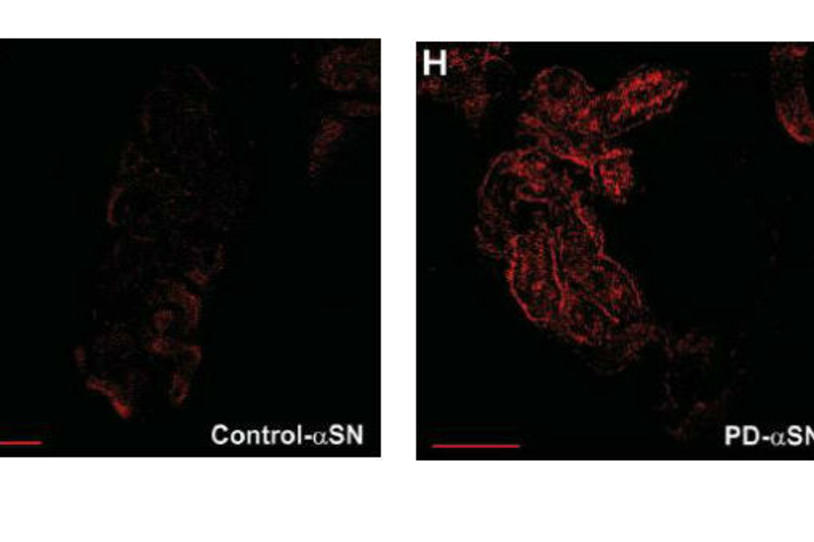
Research has found more alpha-synuclein in the skin of PD patients (right) compared to controls. Image courtesy of Neurology
Reesearchers from Beth Israel Deaconess Medical Center in Boston published a paper in Neurology in late 2013 on their findings that alpha-synuclein levels in nerve cells found in the skin differed between Parkinson’s patients and healthy controls. We talked to Roy Freeman, MD, lead author on the paper, about his team’s study and what this could mean in the pursuit of a Parkinson’s biomarker. Biomarkers are objective measures of disease risk, presence or progression that, as Roy tells us, could have many implications for the field.
The Michael J. Fox Foundation recently funded Roy to continue his work looking at alpha-synuclein levels in the skin. Here we talk to him about his research thus far and next steps.
MJFF: Let’s start high-level: How can a biomarker of Parkinson’s disease help patients?
RF: First, biomarkers can improve diagnostic accuracy. Even in the best of hands, the diagnosis of Parkinson’s disease (PD) may be inaccurate about 20 percent of the time. Second, an accurate and sensitive biomarker could promote early diagnosis. If, for example, we had an effective disease-modifying drug, the sooner one could start treatment the better. Also, in clinical trials, cohort homogeneity is desirable, and a biomarker would help with this. Finally, a biomarker could objectively define disease progression and the response to a disease-modifying therapy.
MJFF: Why are you looking at the skin for a Parkinson’s biomarker?
RF: The skin is remarkable in that it contains many components of the nervous system [Parkinson’s is a disease of the nervous system]. There are, of course, sensory nerves but also several kinds of autonomic nerves: those that innervate the sweat glands, those that innervate the blood vessels and those that innervate the piloerector muscles: the muscles that make your hair stand on end when you’re anxious, scared or cold. Also, the skin is easily accessible. One can access these nerves by doing a minimally invasive skin biopsy, easily repeated time and time again. This allows us to examine disease pathology in living humans as opposed to in a model.
So that’s why the skin. But why look for alpha-synuclein in the skin? Over the past decade there has been growing interest in the non-motor symptoms of Parkinson’s disease, and many of those involve structures outside of the central nervous system (CNS). Functions affected by PD like blood pressure, bladder and bowel control all involve autonomic territories outside the CNS. So we thought with the combination of autonomic involvement in PD, the presence of autonomic nerves in the skin and the accessibility of the skin, why not look for the hallmark Parkinson’s pathology — alpha-synuclein — there?
MJFF: Tell us about your study recently published in Neurology. What did you find?
RF: I view our study as an important first step. We studied 20 patients with Parkinson’s disease and 14 healthy controls, and we looked for the presence of alpha-synuclein in the skin — in sensory nerves and autonomic nerves. We found higher alpha-synuclein levels within the autonomic nerves innervating the sweat glands and piloerector muscles of PD patients. We did not find such in the sensory nerves.
We found also that there is a relationship between the amount of alpha-synuclein deposition in these nerves and Parkinson’s disease severity measured using the Hoehn and Yahr scale scores. There is also a relationship between alpha-synuclein deposition and physiological measures of autonomic function (e.g. blood pressure, heart rate).
MJFF: What might this pathology tell us about Parkinson’s disease?
RF: Well, that’s the next step. We need to look at subjects across a wide array of disease stages including very early in the course of illness. Of particular interest are individuals with pre-motor risk factors for Parkinson’s disease. We then need to look at the disease longitudinally and see what happens to these measures over time and whether the measures can predict the rate of progression.
MJFF: Our Foundation has recently funded some of your additional work in this area. What are the aims of that research?
RF: The Michael J. Fox Foundation has funded two sub-studies. In one study, we will look at the relationship between what we’ve seen in the skin in living subjects with clinically diagnosed Parkinson’s disease and post-mortem tissue from individuals who have a confirmed autopsy diagnosis of Parkinson’s. We’re taking advantage of the Banner Health Institute Brain Program, from whom we will receive autopsy material. This will allow us to determine whether confirmed Parkinson’s patients — defined as those with alpha-synuclein aggregates in the brain — show these same or similar levels of alpha-synuclein in the skin.
In the second sub-study, we want to look at phosphorylated alpha-synuclein. Alpha-synuclein is a normal protein found in healthy individuals, but alpha-synuclein seen in disease state is usually in the phosphorylated form. [Phosphorylation is a process that turns a protein enzyme on or off, altering its function.] The antibody that we used in our study measured total alpha-synuclein, and we want to see what proportion of that total is actually phosphorylated. We suspect quite a bit.
MJFF: What could your findings mean for people living with PD and those who might be at risk?
RF: At this point, we should regard this study as a first step. The study must be replicated by others. More patients must be studied and patients with other parkinsonian syndromes and other neurodegenerative diseases must be studied, too. But, if things proceed as we hope, the goal of having an accessible, reliable, repeatable biomarker will be attained — a biomarker that can confirm the clinical diagnosis, that will predict disease progression, and that will change when modified by a drug. That would be the ideal, but this is the first step. A substantial amount of work needs to be done by us and by others to reach that goal.
